Richard Bertram
Professor, Department of Mathematics
Graduate Faculty Member, Institute
of Molecular Biophysics
Graduate Faculty Member, Program
in Neuroscience
Florida State University

Photo credit: Devin Bittner
Academic Appointments and Honors



- Fellow of the Society for Mathematical Biology, awarded 2023 SMB Fellow Awardees
- Tam Family Professor of Mathematics, 2019-2030
- Distinguished Research Professor, awarded 2019 DRP Awardees
- Marion Bradley Brennan Professor of Mathematics, 2015-2018
- Director of Biomathematics , FSU, 2010-present
- Professor of Biomathematics, FSU, 2009-present
- Graduate Faculty Member, Molecular Biophysics Program, FSU, 2001-present
- Graduate Faculty Member, Neuroscience Program, FSU, 2005-present
Current Funding


- NSF DMS 2324962: Three-year award for "New Approaches for Interpreting Neural Responses to Behaviorally-Relevant Sensory Stimuli", Richard Bertram (PI), Tom Needham, Martin Bauer, Roberto Vincis, 2023-2026.
My Book on Neural Dynamics

Buy the book
Professional Service



- Advisory Board member, Mathematical Biosciences (since 2024).
- Editorial Board member, Bulletin of Mathematical Biology (since 2016).
- Editorial Board member, Biophysical Journal (since 2023).
- Editorial Board member, SIAM book series on Mathematical Neuroscience (since 2024).
- Editorial Board member, SIAM Journal on Life Sciences (since 2025).
Current Research Interests
Activity of Pancreatic Beta-Cells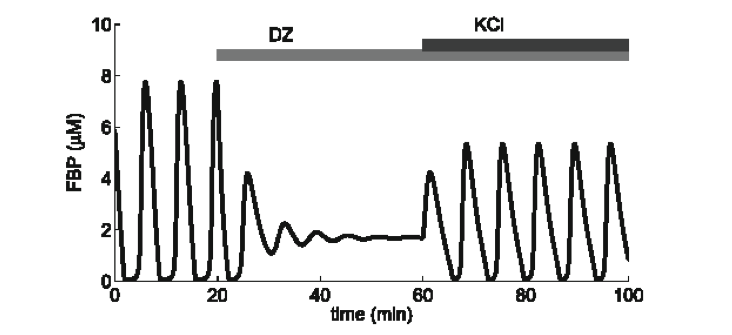
Pancreatic beta-cells are located in islets of Langerhans in the pancreas and are the only cells in the body that secrete insulin,
a hormone that is necessary for the uptake of glucose by other cells. Defects in beta-cell functioning lead to diabetes, which can result
in death if not treated. The release of insulin is controlled by many physiological variables, including the cell's electrical activity,
calcium, and nucleotide concentrations. I work in the development and analysis of mathematical models of beta-cell activity as well as
potential methods for islet syncrhonization.
The Neural Basis of Chemosensing

Humans and other animals sense their environment in a number of ways, including responding to molecules in the air or on the tongue. This
This sensory mechanism is referred to as chemosensing. I collaborate with two experimental labs and a number of fellow mathematicians to use
mathematical modeling and data analysis to understand how taste information is coded in the olfactory bulb and how odor information is coded
in the gustatory cortex of the brain.
Bursting Oscillations in Excitable Cells
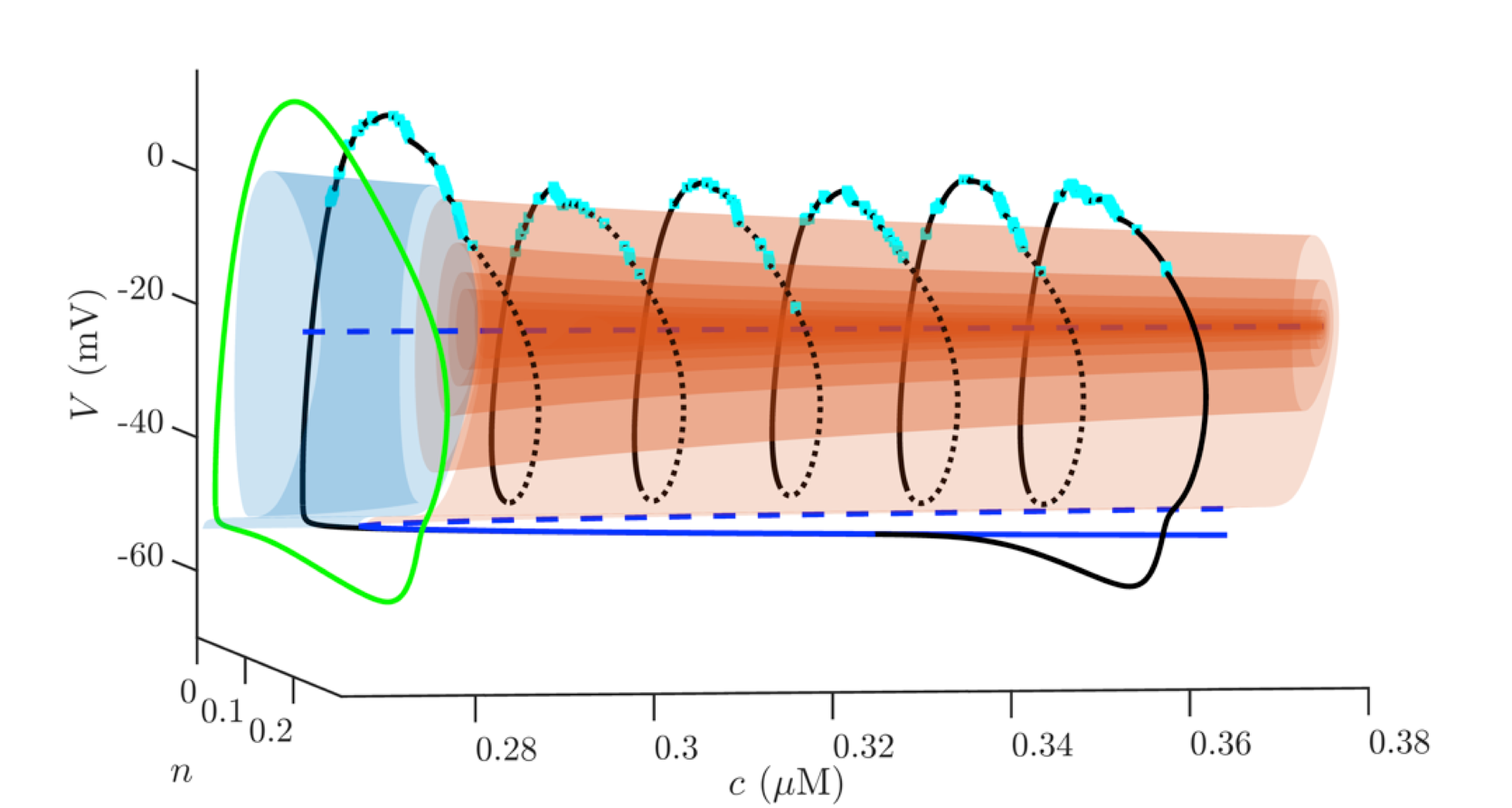
In nerve cells, information is transmitted through electrical impulses. Electrical impulses also cause muscles to contract and endocrine cells
to secrete hormones. Quite often, impulses are generated as high-frequency bursts, followed by periods of quiescence. This is particularly true in
endocrine cells such as pancreatic beta-cells and pituitary cells. I am interested both in the dynamics of bursting (a mathematical topic) and in
the mechanisms by which different cells generate periodic bursts of impulses (a biological topic).
Biological Networks

Many biological systems can be described as networks of interacting parts. I study networks of neurons and hormone-secreting endocrine cells, as well
as gene transcription networks. This research uses techniques from network science, statistics, and data science. Application areas include the olfactory
system, the gustatory system, hypothalamic neurons, and the study of the disease sepsis.
Publications
Neural ModelsPancreatic Beta-Cells
Synaptic Transmission
Structural Biology
Hypothalamus and Pituitary
Neural Basis of Birdsong
Bursting Analysis
Cardiac Models
Other Topics
Lab Members
Students and CollaboratorsComputer Software
Github repositoriesStructural Biology
Hypothalamus and Pituitary
Synaptic Transmission
Pancreatic Islet
Neuron
Birdsong
Bursting Analysis
Cardiac Models
Other Topics
Biomathematics program
VideoBiophysics group
SCUBAMusic and Math
VideosFull CV
Address
Prof. Richard Bertram
Department of Mathematics
Florida State University
Tallahassee, Fl 32306
e-mail: rbertram@fsu.edu